CRACK IT Challenge
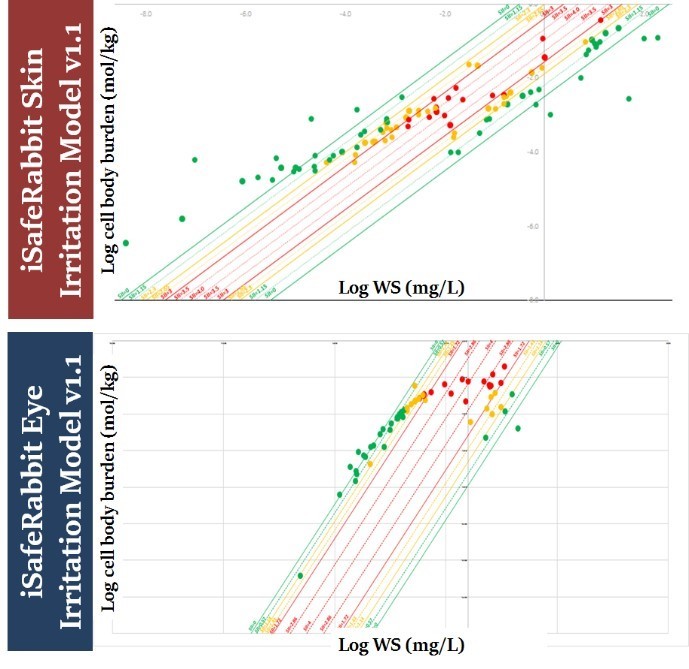
QSARs Mix: Development of high accuracy QSARs for mixtures to bridge the data gaps in skin and eye irritation

At a glance
Completed
Award date
September 2014 - March 2016
Contract amount
£96,500
Contractor(s)
Sponsor(s)