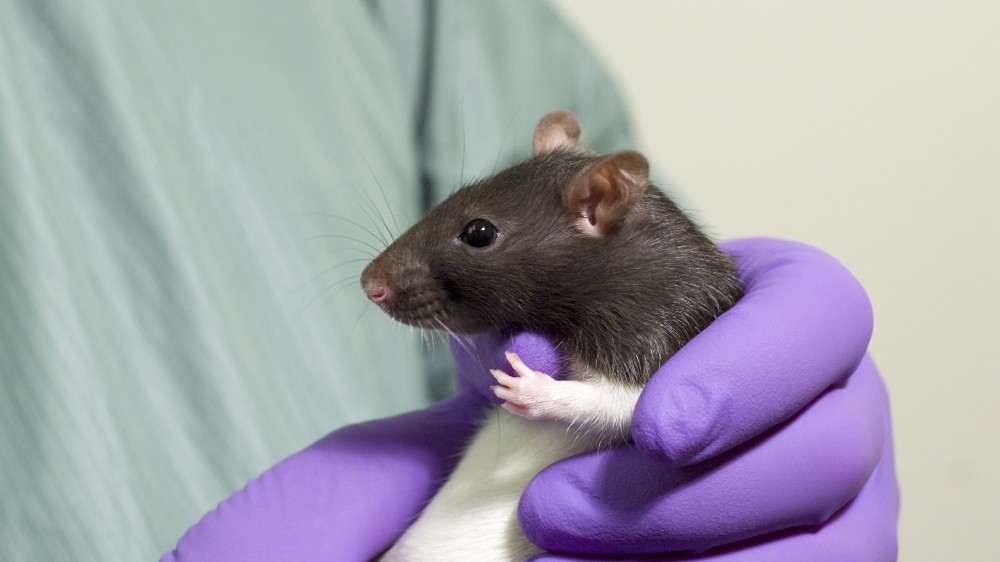
Key results and recommendations from an EPAA/MEB/NC3Rs collaboration focused on chronic toxicity studies for monoclonal antibodies (mAbs).

Key results and recommendations from an EPAA/MEB/NC3Rs collaboration focused on chronic toxicity studies for monoclonal antibodies (mAbs).

A webinar to highlight available opportunities to minimising non-human primate use in drug development.

Opportunities to reduce the use of non-human primates in toxicology programmes.

A webinar to raise awareness of the microsampling technique and encourage wider adoption.
A series of workshops as part of our project to implement the 3Rs in WHO guidelines.
Webinar showcasing ongoing and novel initiatives that replace, reduce and/or refine fish acute toxicity studies.

Providing an evidence base to refine and reduce the use of vertebrates in (regulatory) ecotoxicology studies

Guidance on reducing or refining animal use for studies with cardiovascular, central nervous system and respiratory functional measurements.

Projects providing guidance to reduce animal use or refine procedures within toxicology studies.
Paper clarifies that the forced swim test is not a regulatory requirement for the development of new antidepressants (NC3Rs & MHRA project).
10 years of CRACK IT webinar: A C. elegans platform to assess the developmental and reproductive toxicity (DART) potential of chemicals.

Bibliography and relevant resources for those working in the field of toxicology and regulatory sciences.

A webinar presented as part of the SETAC SciCon 2020.

Examples of study designs that may be used for microsampling from either main study animals or from satellite groups of animals.